
The Orsiro™ Mission™ Sirolimus-Eluting Coronary Stent System is a balloon-expandable stent pre-mounted on a rapid-exchange Percutaneous Transluminal Coronary Angioplasty (PTCA) catheter delivery system.

Flexible shaft
for trackability1,2

designed for biocompatibility1

for pushability1,2


Deep embedding
for crossability

aUltrathin struts
for early endothelialization3,4

Flexible shaft
for trackability

Bioabsorbable coating
with controlled drug release3

Passive coating
designed for biocompatibility1

Enhanced force transmission
for pushability1,2

Deep embedding
for crossability1,2

for early endothelialization3,4

Bioabsorbable coating
with controlled drug release3


Dual-coating
on shaft engineered for limited friction

Ergonomic hub
with kink resistance

on shaft engineered for limited friction

Ergonomic hub
with kink resistance1


Orsiro™ Mission™ DES
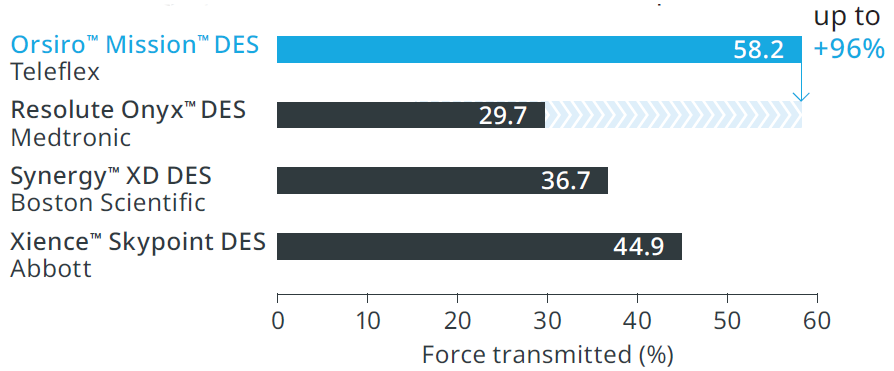
Transmitting up to 96% more force from hub to tip.

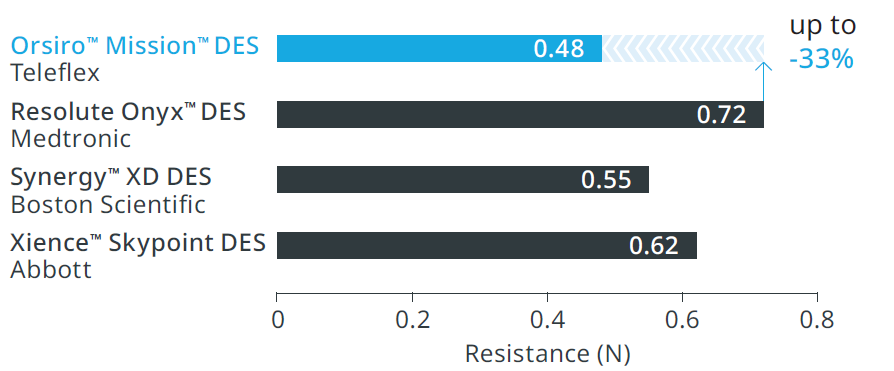
Up to 33% less force needed to follow the path to the lesion.

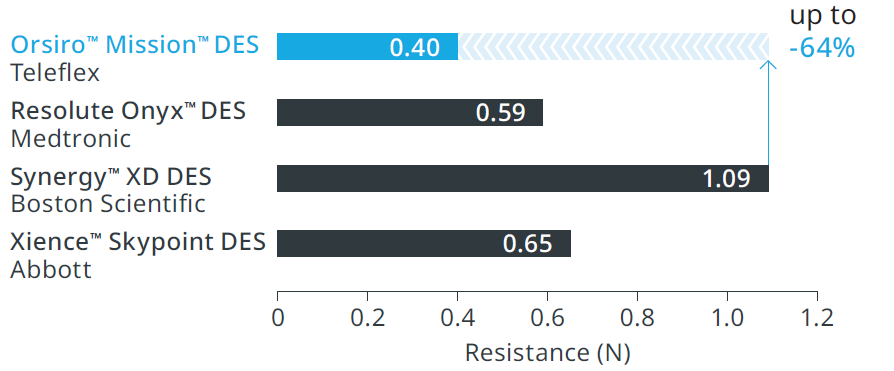

Orsiro™ Mission™ DES
30 days3
>80%
n = 589
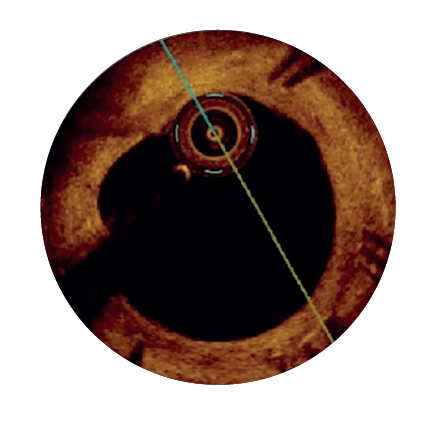
Strut coverage
90 days3
>97%
n = 874
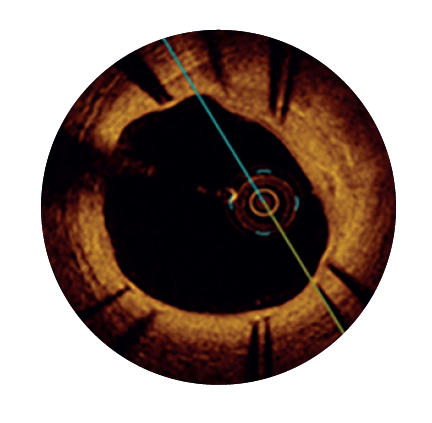
Strut coverage
180 days3
>98%
n = 1,130



Orsiro™ Mission™ DES



| Study Names | Study Type | Patients | Status | Primary Endpoint |
|---|---|---|---|---|
| TAGLIERI et al.12 | Network Meta-Analysis | 99,039 | - | Target Lesion Failure (TLF) at 12 months and the longest follow-up (FU) available |
| BIOFLOW-V11 | Randomized Controlled Trial (RCT) |
1,334 | Completed 60-month FU available |
TLF at 12 months |
| BIO-RESORT8 | RCT | 3,514 | Completed 60-month FU available |
Target Vessel Failure (TVF) at 12 months |
| BIONYX13 | RCT | 2,488 | Completed 36-month FU available |
TVF at 12 months |
| BIOSCIENCE14 | RCT | 2,119 | Completed 60-month FU available |
TLF at 12 months |
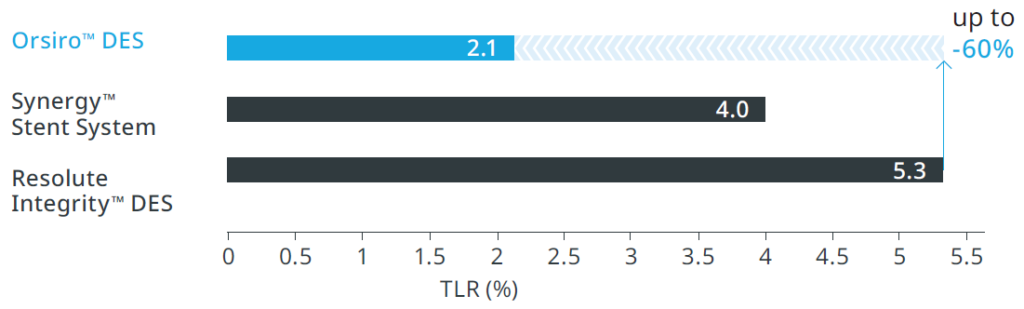

BIOFLOW-V (n = 1,334)




Orsiro™ Mission™ is indicated for improving coronary luminal diameter in patients, including those with diabetes mellitus, with symptomatic heart disease, stable angina, unstable angina, non-ST-elevation myocardial infarction or documented silent ischemia due to atherosclerotic lesions in the native coronary arteries with a reference vessel diameter of 2.25 mm to 4.0 mm and a lesion length of ≤ 36 mm.
| STENT | |
|---|---|
| Stent material | Cobalt chromium, L-605 |
| Strut thickness |
ø 2.25-3.0 mm: 60 µm (0.0024”); ø 3.50-4.0 mm: 80 µm (0.0031”) |
| Passive coating | proBIO™ Coating (Amorphous Silicon Carbide) |
| Active coating | BIOlute™ Coating bioabsorbable Poly-L-Lactide (PLLA) eluting a limus drug |
| Drug dose | 1.4 µg/mm2 |
| DELIVERY SYSTEM | |
|---|---|
| Catheter type | Fast exchange |
| Recommended guide catheter | 5F (min. I.D. 0.056”) |
| Guide wire diameter | 0.014” (0.36 mm) |
| Usable catheter length | 140 cm |
| Balloon material | Semi crystalline polymer |
| Coating (Distal shaft) | Hydrophilic |
| Coating (Proximal shaft) | Hydrophobic |
| Marker bands | Two platinum-iridium markers |
| Proximal shaft diameter | 2.0F |
| Distal shaft diameter | 2.7F: ø 2.25-3.0 mm; 2.9F: ø 3.5-4.0 mm |
| Nominal pressure (NP) | 10 atm |
| Rated burst pressure (RBP) | 16 atm |
| STENT ø | STENT LENGTH | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 9 mm | 13 mm | 15 mm | 18 mm | 22 mm | 26 mm | 30 mm | 35 mm | 40 mm | |
| 2.25 mm | 453925 | 453931 | 453937 | 453943 | 453949 | 453955 | 453961 | ||
| 2.50 mm | 453926 | 453932 | 453938 | 453944 | 453950 | 453956 | 453962 | 453968 | 453974 |
| 2.75 mm | 453927 | 453933 | 453939 | 453945 | 453951 | 453957 | 453963 | 453969 | 453975 |
| 3.00 mm | 453928 | 453934 | 453940 | 453946 | 453952 | 453958 | 453964 | 453970 | 453976 |
| 3.50 mm | 453929 | 453935 | 453941 | 453947 | 453953 | 453959 | 453965 | 453971 | 453977 |
| 4.00 mm | 453930 | 453936 | 453942 | 453948 | 453954 | 453960 | 453966 | 453972 | 453978 |
* Nominal strut thickness for size ø 2.25 – 3.0 mm, mean diameter 62 μm.
** Nominal strut thickness for size ø 2.25 – 2.75 mm.
aUltrathin struts DESs have struts thinner than 70μm.
REFERENCES:
1. Data on file at Teleflex.
2. Testing conducted by the Institute for Implant Technology and Biomaterials and paid for by BIOTRONIK Vascular Intervention, now Teleflex. Test data may not be indicative of clinical performance.
3. Secco, Gioel Gabrio, et al. “Time-related changes in neointimal tissue coverage of a novel Sirolimus eluting stent: Serial observations with optical coherence tomography.” Cardiovascular Revascularization Medicine 2016 17(1): 38-43. Clinical study data reflect results obtained using the Orsiro™ DES and a patient population that was not limited to the authorized indications for use in all regions.
4. Leone, A.; Simonetti, F.; Avvedimento, M.; Angellotti, D.; Immobile Molaro, M.; Franzone, A.; Esposito, G.; Piccolo, R. Ultrathin Struts Drug-Eluting Stents: A State-of-the-Art Review. J. Pers. Med. 2022, 12, 1378. https://doi.org/10.3390/jpm12091378.
5. https://www.cardiovascular.abbott/us/en/hcp/products/percutaneous-coronary-intervention/xience-family/xience-skypoint/ordering-information.html.
6. https://www.medtronic.com/en-us/healthcare-professionals/products/cardiovascular/coronary-stents/resolute-onyx-drug-eluting-stent.html#:~:text=Table_title:%20Product%20specifications%20Table_content:%20header:%20%7C%20Stent,with%20core%20wire%20technology:%20Platinum%20iridium%20%7C.
7. https://www.bostonscientific.com/content/dam/bostonscientific/Interventional%20Cardiology/portfolio-group/Stents/Synergy/resource-center/SYNERGY-Product-Spec-Sheet.pdf#:~:text=4.00%20mm:%200.0032%22%20(81%C2%B5m)%20*Measurement%20applies%20to%20the%202.50%20mm%20diameter%20stent%20system.
8. von Birgelen C, et al. Very thin strut biodegradable polymer everolimus-eluting and sirolimus-eluting stents versus durable polymer zotarolimus-eluting stents in allcomers with coronary artery disease (BIO-RESORT): a three-arm, randomised, non-inferiority trial. The Lancet. 2016 Nov 26;388(10060):2607–2617. doi:10.1016/S0140-6736(16)31920-1. Study sponsored equally by BIOTRONIK Vascular Intervention, now Teleflex, Boston Scientific, and Medtronic. Clinical study data reflect results obtained using the Orsiro™ DES.
9. Buiten RA, Ploumen EH, Zocca P, et al. Thin, Very Thin, or Ultrathin Strut Biodegradeable or Durable Polymer-Coated Drug-Eluting Stents: 3-Year Outcomes of BIO-RESORT. JACC Cardiovasc Interv. 2019;12(17):1650-1660. doi: 10.1016/j.jcin.2019.04.054. Study sponsored equally by BIOTRONIK Vascular Intervention, now Teleflex, Boston Scientific, and Medtronic. Clinical study data reflect results obtained using the Orsiro™ DES.
10. O’Brien BJ et al. A platinum-chromium steel for cardiovascular stents. Biomaterials. 2010; 31(14): 3755-61.
11. Kandzari DE, Mauri L, Koolen JJ, Massaro JM, Doros G, Garcia-Garcia HM, et al. Ultrathin, bioresorbable polymer sirolimus-eluting stents versus thin, durable polymer everolimus-eluting stents in patients undergoing coronary revascularization (BIOFLOW V): a randomized trial. Lancet. 2017 Oct 21;390(10105):1843-1852. doi: 10.1016/S0140-6736(17)32249-3. Epub 2017 Aug 26. Erratum in: Lancet. 2017 Oct 21;390(10105):1832. Study sponsored by BIOTRONIK Vascular Intervention, now Teleflex. Clinical study data reflect results obtained using the Orsiro™ DES.
12. Taglieri N, et al. Target lesion failure with current drug-eluting stents: evidence from a comprehensive network meta-analysis. JACC Cardiovasc Interv. 2020;13(24):2868–2878.
13. von Birgelen, Clemens et al. Thin composite wire strut, durable polymer-coated (Resolute Onyx) versus ultrathin cobalt–chromium strut, bioresorbable polymer-coated (Orsiro) drug-eluting stents in allcomers with coronary artery disease (BIONYX): an international, single-blind, randomised non-inferiority trial. The Lancet, Volume 392, Issue 10154, 1235 – 1245.
14. Pilgrim, Thomas et al. Ultrathin strut biodegradable polymer sirolimus-eluting stent versus durable polymer everolimus-eluting stent for percutaneous coronary revascularisation (BIOSCIENCE): a randomised, single-blind, non-inferiority trial. The Lancet, Volume 384, Issue 9960, 2111 – 2122.
INDICATIONS FOR USE:
Orsiro Mission is indicated for improving coronary luminal diameter in patients, including those with diabetes mellitus, with symptomatic heart disease, stable angina, unstable angina, non-ST elevation myocardial infarction or documented silent ischemia due to atherosclerotic lesions in the native coronary arteries with a reference vessel diameter of 2.25 mm to 4.0 mm and a lesion length of ≤ 36 mm.
CONTRAINDICATIONS:
A known hypersensitivity or allergy to the stent and/or stent coating materials such as amorphous silicon carbide, PLLA polymer, L-605 cobalt chromium alloy (including the major elements cobalt, chromium, tungsten and nickel), sirolimus or its derivatives.
Coronary artery stenting is contraindicated for use in the following patients:
Patients who have contraindications for antiplatelet and/or anticoagulation therapy
Patients who are judged to have a lesion that would be likely to prevent complete inflation of an angioplasty balloon or proper placement of the stent or delivery device
WARNINGS:
Failure to abide by the warnings might result in damage to the device e.g. the coating, which may necessitate intervention or result in serious adverse events.
This device is designed and intended for single use only. Do not resterilize and/or reuse this device. Reuse of a single-use device creates a potential risk of patient or user infections. Contamination of the device may lead to injury, illness, or death of the patient. Cleaning, disinfection, and sterilization may compromise essential material and design characteristics leading to device failure.
Do not use the stent system if the outer package and/or inner package are/is damaged or opened, or if any of the provided information is obscured or damaged.
Do not use the device after the ‘Use by’ date indicated on the label.
Do not attempt to remove or readjust the stent on the delivery system as it may damage the stent, polymer system and/or lead to stent embolization. The stent must not be removed from its dedicated delivery system and placed on another balloon catheter.
Do not expose the stent system to organic solvents e.g. alcohol.
Do not expose and/or suspend the stent within any liquid solution in the sterile field prior to preparation and insertion as the drug carrier coating may be susceptible to damage or premature drug elution.
Administration of appropriate anticoagulant, antiplatelet and vasodilation therapy is critical to successful stent implantation (refer to the sections of the Instructions for Use).
Manipulate the stent system under angiographic guidance when it is in the body.
Use stents with a similar composition when multiple stents are required to treat the lesion as the risk of corrosion increases when stents of dissimilar metals contact one another.
Do not exceed the original diameter of the vessel proximal and distal to the lesion when inflating the balloon to reduce the potential for vessel damage.
Balloon pressure should not exceed the Rated Burst Pressure (RBP). Use of a pressure-monitoring device is mandatory to prevent over-pressurization.
Use only an appropriate balloon inflation medium (e.g. 50:50 mixture by volume of contrast medium and saline). Never use air or any gaseous medium to inflate the balloon.
Subsequent restenosis may require repeat dilation of the arterial segment containing the stent. The long-term outcomes following repeat dilation of endothelialized stents are unknown.
Use outside of the specified indications is prohibited. The use of DES in patients and lesions outside the labeled indications may have an increased risk of adverse events, including stent thrombosis, stent embolization, myocardial infarction (MI), or death.
PRECAUTIONS:
This device should only be used in medical facilities by physicians who are adequately trained and experienced in performing vascular interventions (including cases of life-threatening complications).
If the stent system was removed prior to expansion, do not re-insert as the stent and/or the delivery system may have been damaged during the initial attempt to cross the lesion or during the withdrawal (refer to the sections of the Instructions for Use).
This device carries an associated risk of thrombosis, vascular complications and/or bleeding events. Therefore, careful selection of patients is crucial (refer to the sections of the Instructions for Use).
The long-term effects of DES and the risks associated with these implants are unknown. The limited availability of long-term clinical data should be considered before making a risk/benefit assessment for the patient prior to implantation.
Stent thrombosis is a low-frequency event that is frequently associated with myocardial infarction (MI) or death. Data from the BIOFLOW clinical trials have been prospectively evaluated and adjudicated using the definition developed by the Academic Research Consortium (ARC) (refer to the sections of the Instructions for Use). The rate of stent thrombosis for Orsiro was low in the BIOFLOW clinical trials and did not differ significantly from expectations for a current generation DES.
The patient’s exposure to the drug and polymer system is directly related to the number of stents and the implanted stent length.
Potential interactions of Orsiro Mission with other DES have not been evaluated and should be avoided whenever possible.
ADVERSE EVENTS:
Potential adverse events associated with percutaneous coronary intervention with Orsiro Mission stent placement include, but are not limited to:
Cardiac events: MI or ischemia, abrupt closure of a coronary artery, restenosis of a treated artery (greater than 50% obstruction), cardiogenic shock, angina, tamponade, perforation or dissection of coronary artery or aorta, cardiac perforation, emergency cardiac surgery, pericardial effusion, and aneurysm formation.
Arrhythmic events: ventricular tachycardia, ventricular fibrillation, atrial fibrillation, and bradycardia.
Stent system events: failure to deliver the stent to the intended site, stent dislodgement from the delivery system, stent misplacement, stent deformation, stent embolization, stent thrombosis or occlusion, stent fracture, stent migration, inadequate apposition or compression of stent/s, inflation difficulties, rupture or pinhole of the delivery system balloon, deflation difficulties, withdrawal difficulties, and embolization of catheter material.
Respiratory events: acute pulmonary edema, congestive heart failure, and respiratory insufficiency or failure.
Vascular events: access site hematoma, hypotension/hypertension, pseudoaneurysm, arteriovenous fistula formation, retroperitoneal hematoma, intramural hematoma, vessel dissection or perforation, restenosis, thrombosis or occlusion, vasospasm, peripheral ischemia, dissection and distal embolization (air, tissue debris and thrombus).
Neurologic events: permanent (stroke) or reversible (TIA) neurologic event, femoral nerve injury, and peripheral nerve injury.
Bleeding events: access site bleeding or hemorrhage, hemorrhage requiring transfusion or other treatment.
Allergic reactions to contrast media, antiplatelets, anticoagulants, amorphous silicon carbide, L-605 cobalt chromium alloy, PLLA polymer matrix, sirolimus or sirolimus derivatives.
Contrast-induced nephropathy.
Death.
Infection and sepsis.
Furthermore, all procedure-related adverse events as described in the national and international guidelines of the respective medical associations apply.
Potential adverse events related to oral administration of sirolimus include, but are not limited to, abnormal liver function tests, anemia, arthralgia, diarrhea, hypercholesterolemia, hypersensitivity (including anaphylactic/anaphylactoid type reactions), hypertriglyceridemia, hypokalemia, infections, interstitial lung disease, thrombocytopenia, leukopenia, lymphoma, and other malignancies.
CAUTION:
Federal Law (U.S.A.) restricts this device to sale or use by or on the order of a physician.
Teleflex, the Teleflex logo, BIOlute, Orsiro, Orsiro Mission, and proBIO are trademarks or registered trademarks of Teleflex Incorporated or its affiliates in the U.S. and/or other countries.
All other trademarks marked with a TM are the property of their respective owners and are solely used for identification purposes and do not imply any affiliation, endorsement, or ownership by Teleflex Incorporated or its affiliates. Please see the Instructions for Use for a complete listing of the indications, contraindications, warnings and precautions. Information in this material is not a substitute for the product Instructions for Use. Not all products may be available in all countries. Please contact your local representative.
© 2026 Teleflex Incorporated. All rights reserved. MC-011823 Rev 0. Revised: 04/2026.